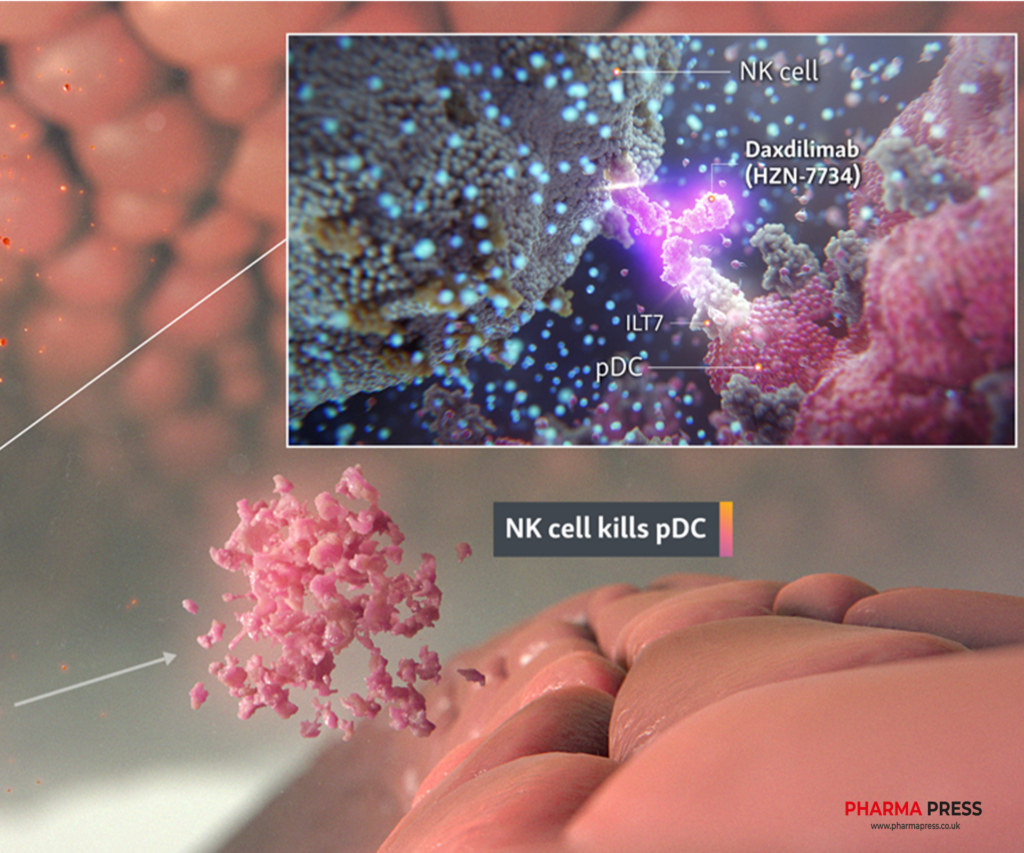
Amgen’s lupus efforts may be about to succeed. Daxdilimab, a monoclonal antibody acquired from Horizon Therapeutics in 2023, hit the primary endpoint in a phase 2 trial for discoid lupus erythematosus.
Daxdilimab significantly reduced disease severity in 72 patients with discoid lupus, an autoimmune condition marked by mostly painless sores on the skin. The phase 2 study is coded as “terminated” on ClinicalTrials.gov because recruitment was stopped early, an Amgen spokesperson told Fierce Biotech, but the trial was in fact completed.
The California company shared the result during a 3 February earnings call, while also touting daxdilimab’s ‘acceptable safety and tolerability profile.’ Another phase 2 trial, in dermatomyositis, had too small a sample size to determine efficacy, Amgen added.
Daxdilimab’s success in discoid lupus follows the antibody’s earlier failure in the more common systemic lupus erythematosus.
In a 2023 phase 2 trial run by Horizon, the molecule failed to beat placebo at improving disease activity. That disappointment came at the same time Amgen’s $27.8 billion buyout of Horizon was on the rocks, with the US Federal Trade Commission suing to block the move before the deal finally went through later in the year.
That same summer, Amgen dropped its own two midstage systemic lupus programs for futility, leaving its lupus pipeline barren.






